Fresenius Kabi Further Expands Oncology Portfolio with Launch of Cyclophosphamide for Injection, USP
February 28, 2024
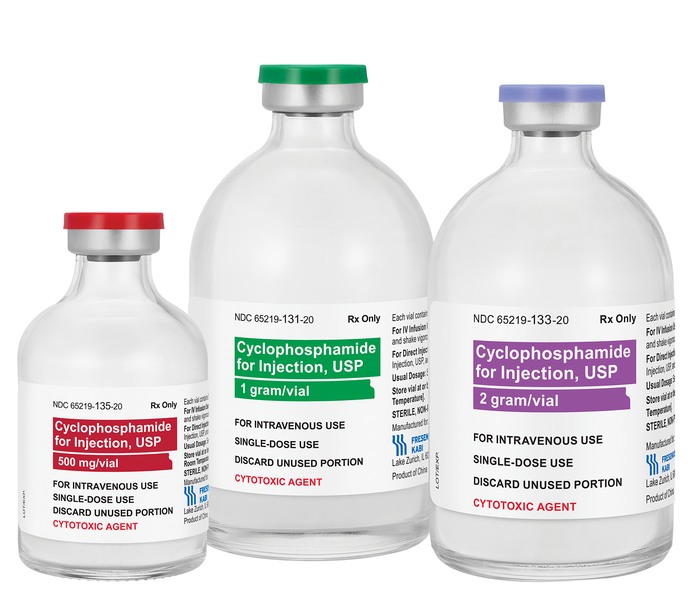
FDA-approved generic provides cost-effective option for treating cancer
LAKE ZURICH, Ill., February 28, 2024 – Fresenius Kabi announced today it has introduced Cyclophosphamide for Injection, USP, a generic substitute for Cytoxan, for use in treating several forms of cancer. Now available in the U.S., Cyclophosphamide for Injection, USP is the newest addition to Fresenius Kabi’s broad portfolio of generic oncology injectables that help make cancer therapies more affordable and accessible.
“The introduction of Cyclophosphamide for Injection adds another critical treatment to our broad and leading oncology portfolio that offers lower-cost options for treating cancer,” said John Ducker, president and CEO of Fresenius Kabi USA.
Supplied as a 500mg, 1g, and 2g single-dose vials of lyophilized powder, the drug can be administered intravenously or orally. Fresenius Kabi offers all lyophilized presentations of cyclophosphamide currently in the market. Cyclophosphamide for Injection, USP is available through distributors or direct from Fresenius Kabi.
About Cyclophosphamide for Injection, USP
Cyclophosphamide for Injection is an alkylating drug indicated for the treatment of:
- Malignant Diseases: malignant lymphomas: Hodgkin's disease, lymphocytic lymphoma, mixed-cell type lymphoma, histiocytic lymphoma, Burkitt's lymphoma; multiple myeloma, leukemias, mycosis fungoides, neuroblastoma, adenocarcinoma of ovary, retinoblastoma, breast carcinoma
- Minimal Change Nephrotic Syndrome in Pediatric Patients: biopsy proven minimal change nephrotic syndrome patients who failed to adequately respond to or are unable to tolerate adrenocorticosteroid therapy
Limitations of Use:
The safety and effectiveness for the treatment of nephrotic syndrome in adults or other renal disease has not been established.
IMPORTANT SAFETY INFORMATION
Cyclophosphamide for Injection is contraindicated in patients with hypersensitivity to cyclophosphamide and with urinary outflow obstruction.
Myelosuppression, Immunosuppression, Bone Marrow Failure and Infections - Severe immunosuppression may lead to serious and sometimes fatal infections. Close hematological monitoring is required.
Urinary Tract and Renal Toxicity - Hemorrhagic cystitis, pyelitis, ureteritis, and hematuria can occur. Exclude or correct any urinary tract obstructions prior to treatment.
Cardiotoxicity - Myocarditis, myopericarditis, pericardial effusion, arrhythmias and congestive heart failure, which may be fatal, have been reported. Monitor patients, especially those with risk factors for cardiotoxicity or pre-existing cardiac disease.
Pulmonary Toxicity - Pneumonitis, pulmonary fibrosis and pulmonary veno-occlusive disease leading to respiratory failure may occur. Monitor patients for signs and symptoms of pulmonary toxicity.
Secondary malignancies have been reported in patients treated with cyclophosphamide-containing regimens.
Veno-occlusive Liver Disease - Fatal outcome can occur.
Embryo-Fetal Toxicity - Can cause fetal harm. Advise female patients of reproductive potential to avoid pregnancy.
Adverse reactions reported most often include neutropenia, febrile neutropenia, fever, alopecia, nausea, vomiting, and diarrhea.
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176, option 5, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Nursing Mothers: Discontinue drug or nursing.
Females and males of reproductive potential: Counsel patients on pregnancy prevention and planning.
Renal Patients: Monitor for toxicity in patients with moderate and severe renal impairment.
This Important Safety Information does not include all the information needed to use Cyclophosphamide for Injection, USP safely and effectively. Please see the full prescribing information for Cyclophosphamide for Injection, USP at www.fresenius-kabi.com/us.
About Fresenius Kabi
Fresenius Kabi (www.fresenius-kabi.com/us) is a global health care company that specializes in injectable medicines, biosimilars, clinical nutrition and technologies for infusion and transfusion. The company’s products and services are used to help care for patients with critical and chronic conditions. The company’s U.S. headquarters is in Lake Zurich, Illinois. The company’s global headquarters is in Bad Homburg, Germany. To learn about U.S. career opportunities at Fresenius Kabi, visit us atwww.fresenius-kabi.com/us/join-us and follow us on LinkedIn and Facebook.
###
