Fresenius Kabi
Epinephrine Injection, USP
1 mg per 1 mL single-dose vial
- Intravenous, intramuscular, subcutaneous, and intraocular use
- FDA AP-rated
- 25 vials per pack
- NDC 63323-696-25
- Download package insert
Wholesale ordering codes:
Cardinal: 5957683
Cencora: 1029-5181
McKesson: 3002607
Morris & Dickson: 441691
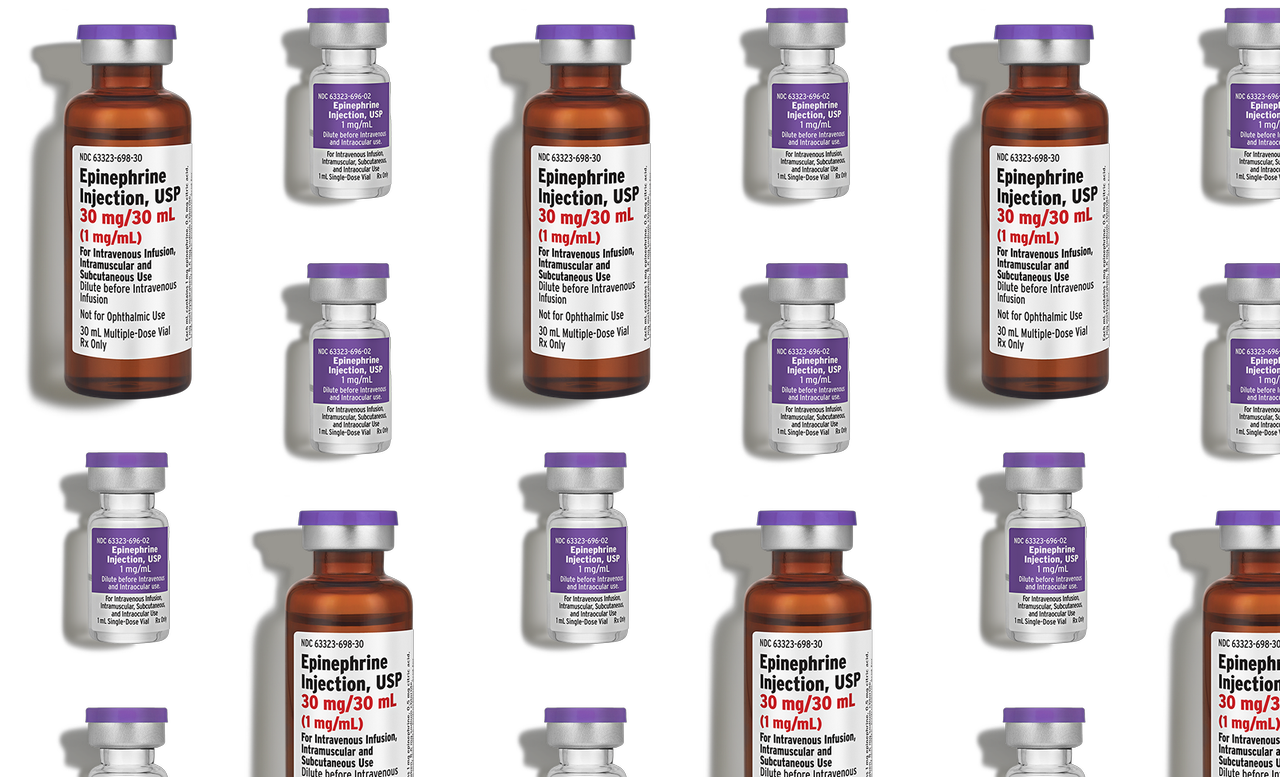
Fresenius Kabi
Epinephrine Injection, USP
30 mg per 30 mL multi-dose vial
- Intravenous, intramuscular, and subcutaneous use
- Pharmaceutically equivalent*
- 1 vial per pack
- NDC 63323-698-30
- J-Code: J0162
- Download package insert
Wholesale ordering codes:
Cardinal: 5983002
Cencora: 1029-9269
McKesson: 3028461
Morris & Dickson: 610154
IMPORTANT SAFETY INFORMATION Select Warnings and Precautions for Epinephrine Injection, USP are as follows:
- Monitor patient for acute severe hypertension.
- Avoid epinephrine extravasation into tissues, which can cause local necrosis.
- Do not inject epinephrine into buttocks, digits, hands, or feet.
- Potential for pulmonary edema when epinephrine is administered intravenously, which may be fatal. Treat with a rapidly acting alpha-adrenergic blocking drug and respiratory support.
- May constrict renal blood vessels and decrease urine formation.
- May induce potentially serious cardiac arrhythmias or aggravate angina pectoris, particularly in patients with underlying heart disease.
- Presence of sulfite in this product should not deter use for treatment of serious allergic or other emergency situations.
Most common adverse reactions to systemically administered epinephrine are headache; anxiety; apprehensiveness; restlessness; tremor; weakness; dizziness; sweating; palpitations; pallor; peripheral coldness; nausea/vomiting; and/or respiratory difficulties. Arrhythmias, including fatal ventricular fibrillation, rapid rises in blood pressure producing cerebral hemorrhage, and angina have occurred.
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176, option 5, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Drugs that counter the pressor effects of epinephrine include alpha blockers, vasodilators such as nitrates, diuretics, antihypertensives, and ergot alkaloids.
- Drugs that potentiate the effects of epinephrine include sympathomimetics, beta blockers, tricyclic antidepressants, MAO inhibitors, COMT inhibitors, clonidine, doxapram, oxytocin, levothyroxine sodium, and certain antihistamines.
- Drugs that increase the arrhythmogenic potential of epinephrine include beta blockers, cyclopropane and halogenated hydrocarbon anesthetics, quinidine, antihistamines, exogenous thyroid hormones, diuretics, and cardiac glycosides. Observe for development of cardiac arrhythmias.
- Potassium-depleting drugs, including corticosteroids, diuretics, and theophylline, potentiate the hypokalemic effects of epinephrine.
Pregnancy: May cause fetal harm.
Elderly patients and pregnant women may be at greater risk of developing adverse reactions when epinephrine is administered parenterally.
INDICATIONS AND USAGE
Epinephrine is a non-selective alpha and beta adrenergic agonist indicated:
- To increase mean arterial blood pressure in adult patients with hypotension associated with septic shock.
- For emergency treatment of allergic reactions (Type 1), including anaphylaxis.
- For induction and maintenance of mydriasis during intraocular surgery.
This Important Safety Information does not include all the information needed to use Epinephrine Injection, USP safely and effectively. For full prescribing information for Epinephrine Injection, USP, please see package insert here.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
- Do not inject into buttocks, digits, hands, or feet.
- Avoid extravasation into tissues, which can cause local necrosis.
- Monitor patient for acute severe hypertension.
- Potential for pulmonary edema, which may be fatal.
- May constrict renal blood vessels and decrease urine formation.
- May induce potentially serious cardiac arrhythmias or aggravate angina pectoris, particularly in patients with underlying heart disease.
- Presence of sulfite in this product should not deter use.
ADVERSE REACTIONS
Most common adverse reactions to systemically administered epinephrine are headache; anxiety; apprehensiveness; restlessness; tremor; weakness; dizziness; sweating; palpitations; pallor; peripheral coldness; nausea/vomiting; and/or respiratory difficulties. Arrhythmias, including fatal ventricular fibrillation, rapid rises in blood pressure producing cerebral hemorrhage, and angina have occurred.
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176, option 5, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Drugs that counter the pressor effects of epinephrine include alpha blockers, vasodilators such as nitrates, diuretics, antihypertensives, ergot alkaloids, and phenothiazine antipsychotics.
- Drugs that potentiate the effects of epinephrine include sympathomimetics, beta blockers, tricyclic antidepressants, MAO inhibitors, COMT inhibitors, clonidine, doxapram, oxytocin.
- Drugs that increase the arrhythmogenic potential of epinephrine include beta blockers, cyclopropane and halogenated hydrocarbon anesthetics, quinidine, antihistamines, exogenous thyroid hormones, diuretics, and cardiac glycosides. Observe for development of cardiac arrhythmias.
- Potassium-depleting drugs, including corticosteroids, diuretics, and theophylline, potentiate the hypokalemic effects of epinephrine.
USE IN SPECIFIC POPULATIONS
Pregnancy: May cause fetal harm.
Elderly patients and pregnant women may be at greater risk of developing adverse reactions when epinephrine is administered parenterally.
INDICATIONS AND USAGE
Epinephrine is a non-selective alpha and beta adrenergic agonist indicated:
- For emergency treatment of allergic reactions (Type 1), including anaphylaxis, in adults and pediatric patients.
- To increase mean arterial blood pressure in adult patients with hypotension associated with septic shock.
This Important Safety Information does not include all the information needed to use Epinephrine Injection, USP safely and effectively. For full prescribing information for Epinephrine Injection, USP, please see package insert here.