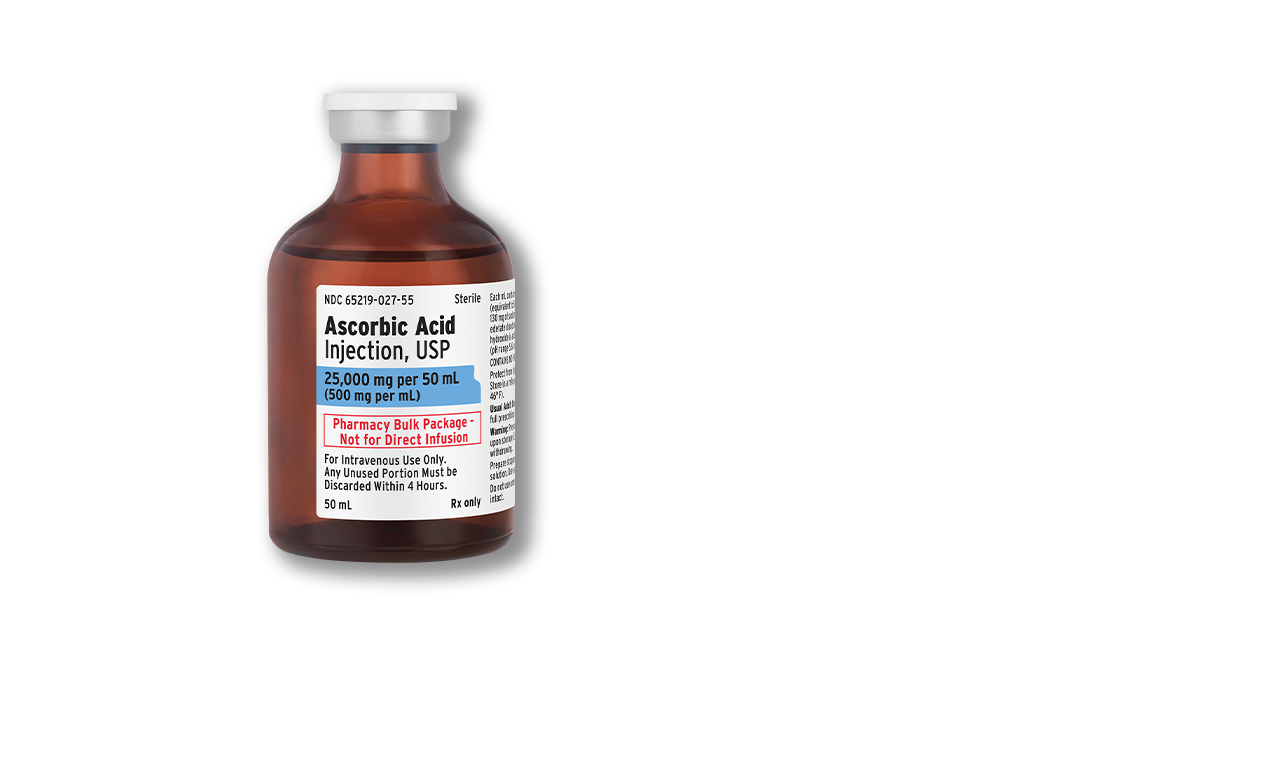
Ascorbic Acid Injection, USP

Download Package Insert
- AP-rated
- 36-month shelf life
- Preservative free
- Container closure not made with natural rubber latex
- Formulated, filled and packaged in the U.S.
More in America: Strengthening Supply Reliability
We know supply continuity matters. That's why Fresenius Kabi's broad U.S. production presence helps ensure consistent availability of Ascorbic Acid Injection, USP.
More in America is Fresenius Kabi’s ongoing commitment to strengthening our U.S. production and supply capabilities. Through expanded capacity, increased safety stock, and investments in automation, Fresenius Kabi is focused on improved reliability for critical medications – so providers can focus on patient care.